Analysis of Corn Stover
Background on Corn Stover
This resource is so large partially because of the amount of corn grown in the US, but also because of the amount of residues produced by the crop; it has been estimated that corn in the US produces 1.7 times more residue than other cereals.
This field harvest residue consists of the stalk (around 48% of the total dry mass), leaves (28%), and husks (8%) of the plant, as well as the cob (15%).
For stover and other straws, the amount of residue that needs to be left on the soil to preserve soil fertility and prevent against soil erosion is an important consideration. This is a complex issue and the field stover requirements will vary according to the location and factors such as climate, soil characteristics, tillage type and crop rotations.
Corn stover can be harvested wet or after drying on the field for a period of time. A dry harvest involves the residue being shredded and raked before being left to field dry and at a later point the stover is windrowed and baled. Since this is a multi-pass operation it can be costly and means that grain harvesting and residue collecting operations cannot be integrated. An alternative is to harvest the product wet in which case the shredding, windrowing and baling can take place on the same day as grain harvest with the first two operations possible from the grain harvester itself. The stover can then be ensiled to prevent storage losses.
The target of many researchers developing integrated harvesting practices is a one-pass system that allows for the simultaneous harvest of grain and stover while also leaving enough residue on the field to preserve soil fertility. There is also work on modifying the header of such an integrated combine so that the relative proportions of the stover fractions (cobs, stalks etc.) can be varied according to the end needs of the biorefinery.
Analysis of Corn Stover at Celignis
Celignis Analytical can determine the following properties of Corn Stover samples:
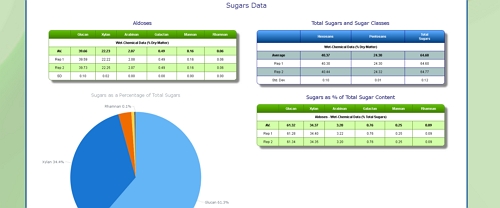
Lignocellulosic Properties of Corn Stover
Cellulose Content of Corn Stover
For example, the cellulose content is typically highest in the stalk, and less in the husks, lower still in the cob, and lowest in the leaves.
Furthermore the composition of these residues will also vary due to the variety of the plant and its productivity.
Click here to see the Celignis Analysis Packages that determine Cellulose Content
Request a QuoteCellulose Content
Hemicellulose Content of Corn Stover
Click here to see the Celignis Analysis Packages that determine Hemicellulose Content
Request a QuoteHemicellulose Content
Lignin Content of Corn Stover
Click here to see the Celignis Analysis Packages that determine Lignin Content
Request a QuoteLignin Content
Starch Content of Corn Stover
Click here to see the Celignis Analysis Packages that determine Starch Content
Request a QuoteStarch Content
Uronic Acid Content of Corn Stover
Click here to see the Celignis Analysis Packages that determine Uronic Acid Content
Request a QuoteUronic Acid Content
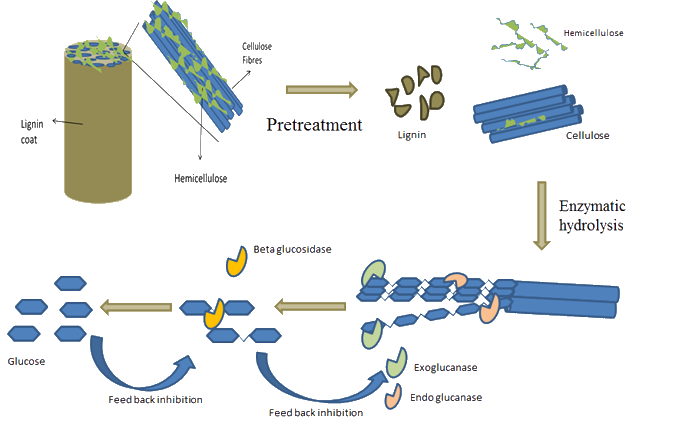
Enzymatic Hydrolysis of Corn Stover
Click here to see the Celignis Analysis Packages that determine Enzymatic Hydrolysis
Request a QuoteEnzymatic Hydrolysis
Bioenergy Properties of Corn Stover
Ash Content of Corn Stover
Click here to see the Celignis Analysis Packages that determine Ash Content
Request a QuoteAsh Content
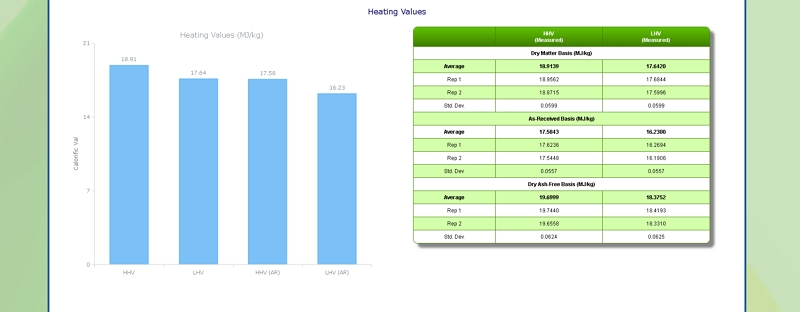
Heating (Calorific) Value of Corn Stover
Click here to see the Celignis Analysis Packages that determine Heating (Calorific) Value
Request a QuoteHeating (Calorific) Value
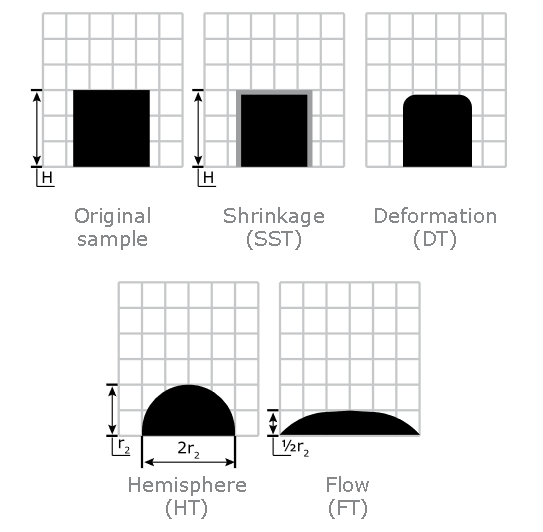
Ash Melting Behaviour of Corn Stover
Ash Shrinkage Starting Temperature (SST) - This occurs when the area of the test piece of Corn Stover ash falls below 95% of the original test piece area.
Ash Deformation Temperature (DT) - The temperature at which the first signs of rounding of the edges of the test piece occurs due to melting.
Ash Hemisphere Temperature (HT) - When the test piece of Corn Stover ash forms a hemisphere (i.e. the height becomes equal to half the base diameter).
Ash Flow Temperature (FT) - The temperature at which the Corn Stover ash is spread out over the supporting tile in a layer, the height of which is half of the test piece at the hemisphere temperature.
Click here to see the Celignis Analysis Packages that determine Ash Melting Behaviour
Request a QuoteAsh Melting Behaviour
Major and Minor Elements in Corn Stover
We can also determine the levels of 13 different minor elements (such as arsenic, copper, and zinc) that may be present in Corn Stover.
Click here to see the Celignis Analysis Packages that determine Major and Minor Elements
Request a QuoteMajor and Minor Elements
Analysis of Corn Stover for Anaerobic Digestion
Biomethane potential (BMP) of Corn Stover
At Celignis we can provide you with crucial data on feedstock suitability for AD as well as on the composition of process residues. For example, we can determine the biomethane potential (BMP) of Corn Stover. The BMP can be considered to be the experimental theoretical maximum amount of methane produced from a feedstock. We moniotor the volume of biogas produced allowing for a cumulative plot over time, accessed via the Celignis Database. Our BMP packages also involve routine analysis of biogas composition (biomethane, carbon dioxide, hydrogen sulphide, ammonia, oxygen). We also provide detailed analysis of the digestate, the residue that remains after a sample has been digested. Our expertise in lignocellulosic analysis can allow for detailed insight regarding the fate of the different biogenic polymers during digestion.
Click here to see the Celignis Analysis Packages that determine BMP
Request a QuoteBMP
Physical Properties of Corn Stover
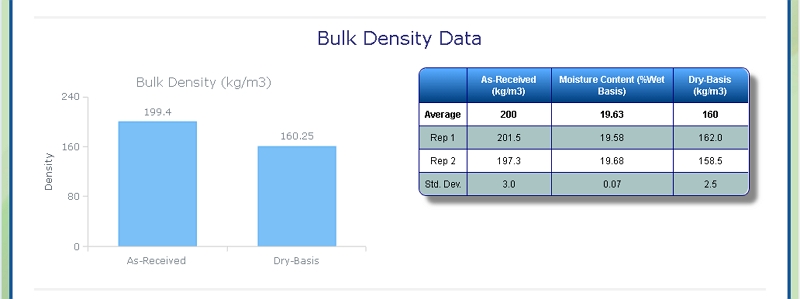
Bulk Density of Corn Stover
At Celignis we can determine the bulk density of biomass samples, including Corn Stover, according to ISO standard 17828 (2015). This method requires the biomass to be in an appropriate form (chips or powder) for density determination.
Click here to see the Celignis Analysis Packages that determine Bulk Density
Request a QuoteBulk Density
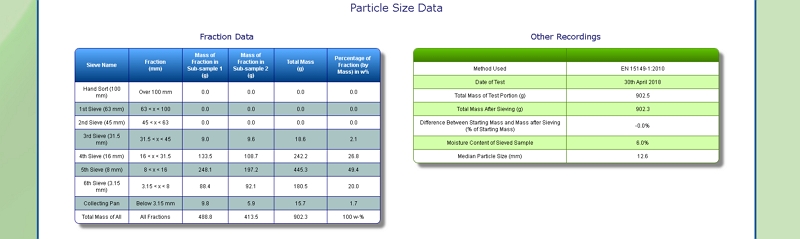
Particle Size of Corn Stover
Our lab is equipped with a Retsch AS 400 sieve shaker. It can accommodate sieves of up to 40 cm diameter, corresponding to a surface area of 1256 square centimetres. This allows us to determine the particle size distribution of a range of samples, including Corn Stover, by following European Standard methods EN 15149- 1:2010 and EN 15149-2:2010.
Click here to see the Celignis Analysis Packages that determine Particle Size
Request a QuoteParticle Size
Fruit juice processing generates large volumes of organic waste, including pomace, retentate, and waste apples, that are a challenge to manage. Anaerobic digestion (AD) allows for conversion of these wastes into biogas; however, their high acidity and low buffering capacity limits AD process stability, leading to reduced methane yield. In this study, co-digestion with manure and lignocellulosic biomass (LCB) was assessed. A five-factor mixture design was used to test different combinations on a bench scale, selected based-on seasonal fruit waste availability. Process performance was assessed based-on methane yield and volatile fatty acids before and after AD. Feedstock mixture representing an off-season blend of 20% pomace, 30% retentate and 50% manure, as well as an in-season blends of 20% waste apples, 30% pomace, 30% retentate, and 20% manure, were found to maximise the biomethane yield. Supplementation with at least 20% manure was essential for fruit waste digestion. Replacing a portion of the fruit waste with lignocellulose in the anaerobic digestion significantly improved the methane yield and prevented an “acid crash”. It was found that 30% LCB and 20% manure supplementation were the minimum required for anaerobic digestion process stability and yield for both in- and off-season fruit harvesting and processing. | ||
Next-generation biofuels from renewable sources have gained interest among research investigators, industrialists, and governments due to major concerns on the volatility of oil prices, climate change, and depletion of oil reserves. Biobutanol has drawn signicant attention as an alternative transportation fuel due to its superior fuel properties over ethanol. e advantages of butanol are its high energy content, better blending with gasoline, less hydroscopic nature, lower volatility, direct use in convention engines, low corrosiveness, etc. Butanol production through (acetone, butanol, and ethanol) ABE fermentation is a well-established process, but it has several drawbacks like feedstock cost, strain degeneration, product toxicity, and low product concentrations. Lignocellulosic biomass is considered as the most abundant, renewable, low-cost feedstock for biofuels. Production of butanol from lignocellulosic biomass is more complicated due to the recalcitrance of feedstock and inhibitors generated during the pretreatment and hydrolysis process. Advanced fermentation and product recovery techniques are being researched to make biobutanol industrially viable. | |