Analysis of Pulp
Background on Pulp
Analysis of Pulp at Celignis
Celignis Analytical can determine the following properties of Pulp samples:
Lignocellulosic Properties of Pulp
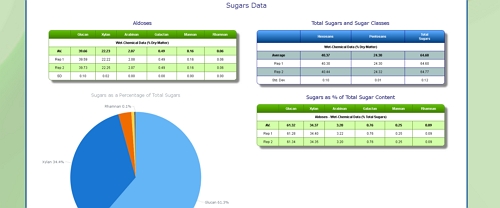
Cellulose Content of Pulp
Click here to see the Celignis Analysis Packages that determine Cellulose Content
Request a QuoteCellulose Content
Hemicellulose Content of Pulp
Click here to see the Celignis Analysis Packages that determine Hemicellulose Content
Request a QuoteHemicellulose Content
Lignin Content of Pulp
Click here to see the Celignis Analysis Packages that determine Lignin Content
Request a QuoteLignin Content
Starch Content of Pulp
Click here to see the Celignis Analysis Packages that determine Starch Content
Request a QuoteStarch Content
Uronic Acid Content of Pulp
Click here to see the Celignis Analysis Packages that determine Uronic Acid Content
Request a QuoteUronic Acid Content
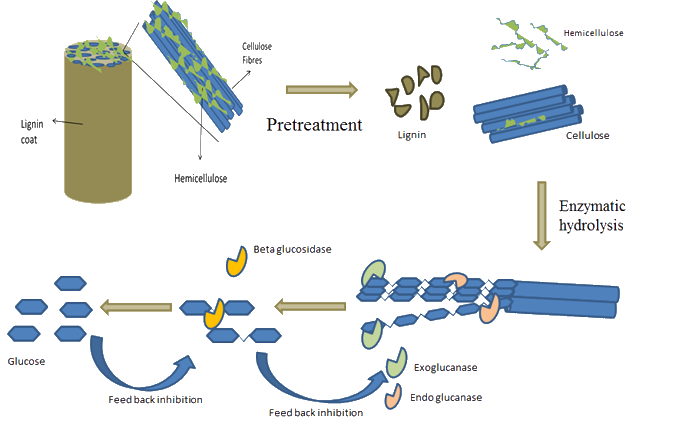
Enzymatic Hydrolysis of Pulp
Click here to see the Celignis Analysis Packages that determine Enzymatic Hydrolysis
Request a QuoteEnzymatic Hydrolysis
Bioenergy Properties of Pulp
Ash Content of Pulp
Click here to see the Celignis Analysis Packages that determine Ash Content
Request a QuoteAsh Content
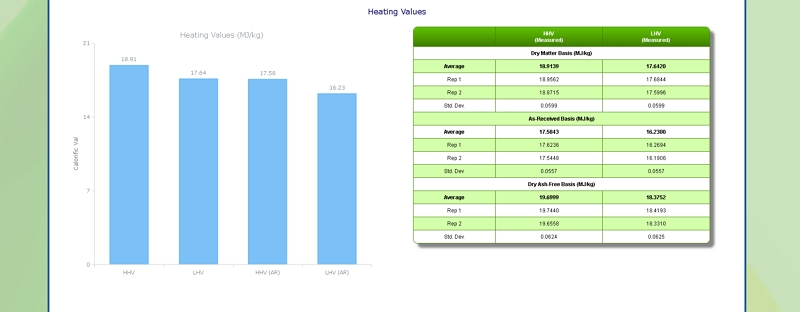
Heating (Calorific) Value of Pulp
Click here to see the Celignis Analysis Packages that determine Heating (Calorific) Value
Request a QuoteHeating (Calorific) Value
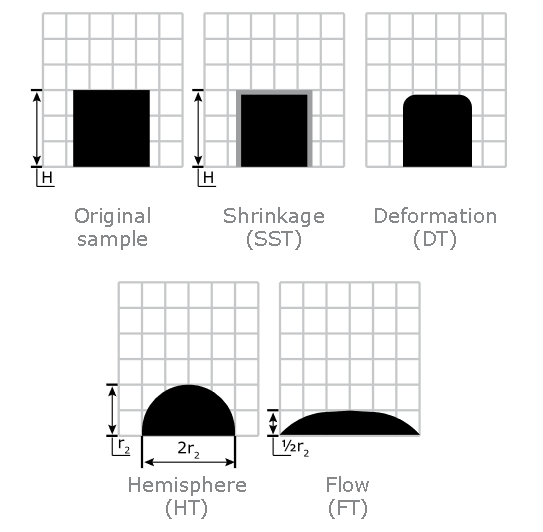
Ash Melting Behaviour of Pulp
Ash Shrinkage Starting Temperature (SST) - This occurs when the area of the test piece of Pulp ash falls below 95% of the original test piece area.
Ash Deformation Temperature (DT) - The temperature at which the first signs of rounding of the edges of the test piece occurs due to melting.
Ash Hemisphere Temperature (HT) - When the test piece of Pulp ash forms a hemisphere (i.e. the height becomes equal to half the base diameter).
Ash Flow Temperature (FT) - The temperature at which the Pulp ash is spread out over the supporting tile in a layer, the height of which is half of the test piece at the hemisphere temperature.
Click here to see the Celignis Analysis Packages that determine Ash Melting Behaviour
Request a QuoteAsh Melting Behaviour
Major and Minor Elements in Pulp
We can also determine the levels of 13 different minor elements (such as arsenic, copper, and zinc) that may be present in Pulp.
Click here to see the Celignis Analysis Packages that determine Major and Minor Elements
Request a QuoteMajor and Minor Elements
Analysis of Pulp for Anaerobic Digestion
Biomethane potential (BMP) of Pulp
At Celignis we can provide you with crucial data on feedstock suitability for AD as well as on the composition of process residues. For example, we can determine the biomethane potential (BMP) of Pulp. The BMP can be considered to be the experimental theoretical maximum amount of methane produced from a feedstock. We moniotor the volume of biogas produced allowing for a cumulative plot over time, accessed via the Celignis Database. Our BMP packages also involve routine analysis of biogas composition (biomethane, carbon dioxide, hydrogen sulphide, ammonia, oxygen). We also provide detailed analysis of the digestate, the residue that remains after a sample has been digested. Our expertise in lignocellulosic analysis can allow for detailed insight regarding the fate of the different biogenic polymers during digestion.
Click here to see the Celignis Analysis Packages that determine BMP
Request a QuoteBMP
Physical Properties of Pulp
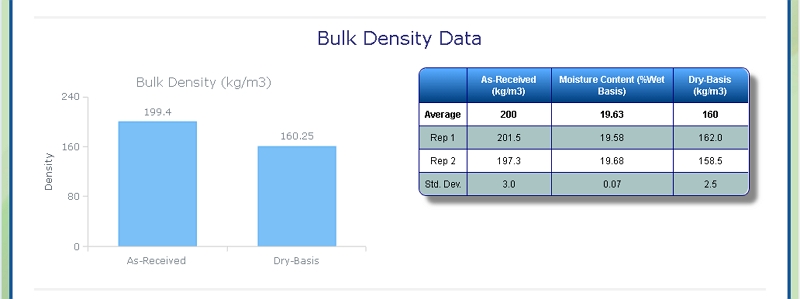
Bulk Density of Pulp
At Celignis we can determine the bulk density of biomass samples, including Pulp, according to ISO standard 17828 (2015). This method requires the biomass to be in an appropriate form (chips or powder) for density determination.
Click here to see the Celignis Analysis Packages that determine Bulk Density
Request a QuoteBulk Density
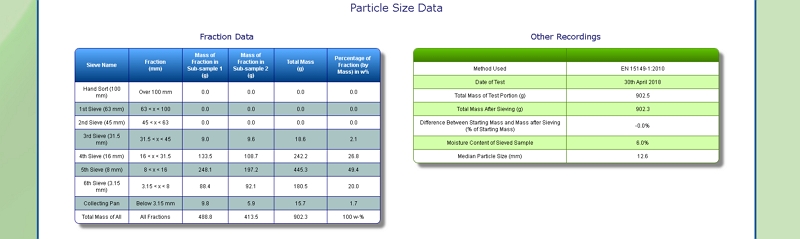
Particle Size of Pulp
Our lab is equipped with a Retsch AS 400 sieve shaker. It can accommodate sieves of up to 40 cm diameter, corresponding to a surface area of 1256 square centimetres. This allows us to determine the particle size distribution of a range of samples, including Pulp, by following European Standard methods EN 15149- 1:2010 and EN 15149-2:2010.
Click here to see the Celignis Analysis Packages that determine Particle Size
Request a QuoteParticle Size
This study evaluated the physicochemical properties and bioenergy potential of three distinct paper sludges: virgin pulp sludge (VP-PS), corrugated cardboard sludge (CR-PS), and tissue and printing paper sludge (TPR-PS). From the experimental runs, VP-PS exhibited the highest ethanol yield (46.8 ± 3.7 g L−1, 87.4% conversion), which can be attributed to its high glucan content and efficient enzymatic hydrolysis. Corrugated cardboard, despite its higher lignin content, demonstrated superior biogas and methane production (94.4 ± 8.3 L kg−1 and 54.4 ± 5.1 L CH4 kg−1 TSfed), likely due to its elevated xylan levels and favorable bulk density. The TPR-PS was characterized by high ash content and showed lower performance for both bioenergy production pathways but displayed improved solid handling due to its higher bulk density and lower water-holding capacity. These results provide a good explanation of the potential bioconversion pathways for the different paper sludge characteristics to maximize bioenergy yield. | ||
Disposal of waste sludges produced in large amounts in the pulp and paper industry imposes significant environmental and economical problems. One strategy to address these issues involves revalorization of paper mill sludges by their application as substrates for microbial production of biotechnologically relevant enzymes. The application of lignocellulolytic enzymes in paper, textile and bioenergy industries is encouraged in order to decrease chemicals and energy consumptions. In the following work, deinking sludge was assessed as a substrate for production of lignocellulases. Based on the results of growth and activity screenings, Pleurotus ostreatus PLAB was chosen as the most promising candidate among 30 tested strains and its secretome was further studied by quantitative enzyme assays and mass spectrometry. While endoglucanase and xylanase activities detected in P. ostreatus secretome produced on deinking sludge were similar to activities of cultures grown on other lignocellulosic substrates, average laccase activity was significantly higher (46?000 U/kg DIS). Mass spectrometry identification of the most prominent proteins in the secretome of the target strain confirmed that significant amounts of different lignin-modifying oxidases were produced on this substrate despite its low lignin content, indicating the presence of other inducible compounds. The findings of this study suggest deinking sludge may represent a good substrate for fungal production of the aforementioned enzymes with broad biotechnological applications, including bioremediation, paper and bioenergy industries. | |
Paper sludge samples collected from recycling mills exhibited high ash content in the range of 54.59%�65.50% and glucose concentrations between 21.97% and 31.11%. Washing the sludge reduced the total ash content to between 10.7% and 19.31% and increased the concentration of glucose, xylose and lignin. Samples were screened for ethanol production and fed-batch simultaneous saccharification and fermentation (SSF) was optimised for the washed samples that resulted in highest and lowest ethanol concentrations. Maximum ethanol concentrations of 57.31 g/L and 47.72 g/L (94.07% and 85.34% of the maximum theoretical yield, respectively) was predicted for high and low fermentative potential samples, respectively, and was experimentally achieved with 1% deviation. A generic set of process conditions were established for the conversion of high ash-containing paper sludge to ethanol. Techno-economic analysis based on three different revenue scenarios, together with Monte Carlo analysis revealed 95% probability of achieving IRR values in excess of 25% at a paper sludge feed rate of 15 t/d. Feed rates of 30 t/d and 50 t/d exhibited a cumulative probability of 100%. This study presents the technical feasibility and economic viability of paper mills expansion towards bioethanol production from paper sludge. | |