Analysis of Foliage
Background on Foliage
Analysis of Foliage at Celignis
Celignis Analytical can determine the following properties of Foliage samples:
Lignocellulosic Properties of Foliage
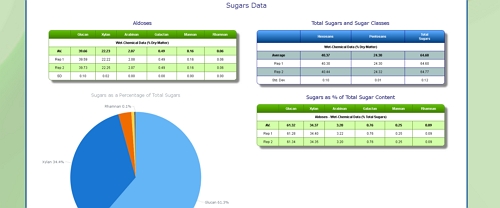
Cellulose Content of Foliage
The chemical composition of foliage can vary significantly, according to the plant. For example, hardwood trees typically have broad leaves whilst softwood trees have needles. Furthermore, whilst hardwoods are deciduous, meaining that their leaves are shed each year, softwood conifers are evergreen.
Foilage from softwoods tends to have a higher cellulose content than foliage from hardwoods, but this will depend on the species in question.
Click here to see the Celignis Analysis Packages that determine Cellulose Content
Request a QuoteCellulose Content
Hemicellulose Content of Foliage
Click here to see the Celignis Analysis Packages that determine Hemicellulose Content
Request a QuoteHemicellulose Content
Lignin Content of Foliage
Click here to see the Celignis Analysis Packages that determine Lignin Content
Request a QuoteLignin Content
Starch Content of Foliage
Click here to see the Celignis Analysis Packages that determine Starch Content
Request a QuoteStarch Content
Uronic Acid Content of Foliage
Click here to see the Celignis Analysis Packages that determine Uronic Acid Content
Request a QuoteUronic Acid Content
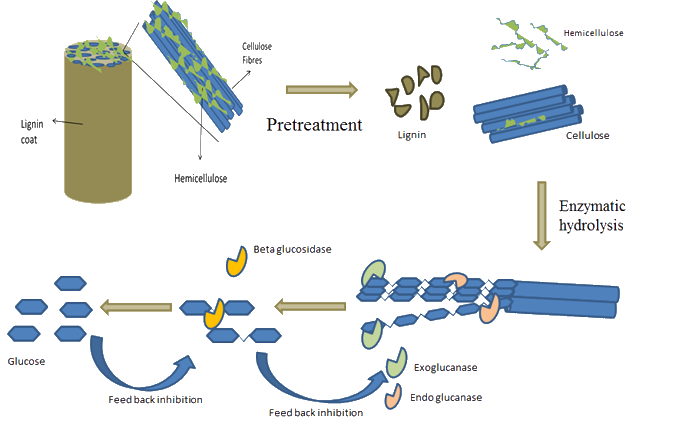
Enzymatic Hydrolysis of Foliage
Click here to see the Celignis Analysis Packages that determine Enzymatic Hydrolysis
Request a QuoteEnzymatic Hydrolysis
Bioenergy Properties of Foliage
Ash Content of Foliage
Click here to see the Celignis Analysis Packages that determine Ash Content
Request a QuoteAsh Content
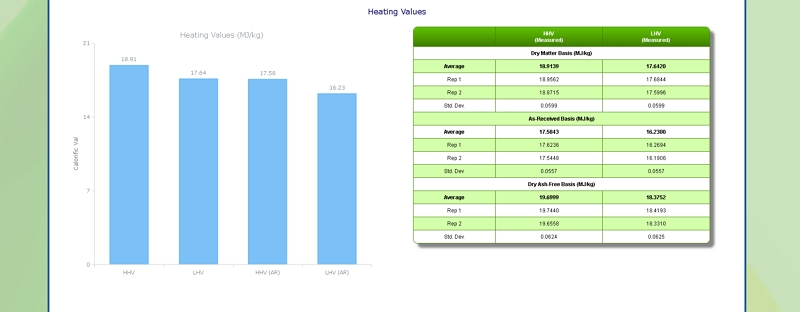
Heating (Calorific) Value of Foliage
Click here to see the Celignis Analysis Packages that determine Heating (Calorific) Value
Request a QuoteHeating (Calorific) Value
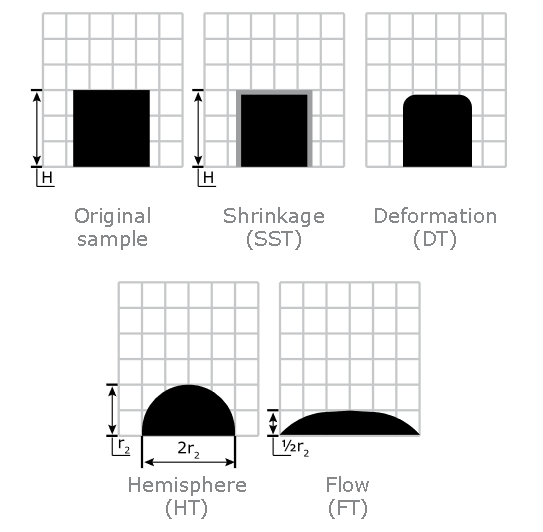
Ash Melting Behaviour of Foliage
Ash Shrinkage Starting Temperature (SST) - This occurs when the area of the test piece of Foliage ash falls below 95% of the original test piece area.
Ash Deformation Temperature (DT) - The temperature at which the first signs of rounding of the edges of the test piece occurs due to melting.
Ash Hemisphere Temperature (HT) - When the test piece of Foliage ash forms a hemisphere (i.e. the height becomes equal to half the base diameter).
Ash Flow Temperature (FT) - The temperature at which the Foliage ash is spread out over the supporting tile in a layer, the height of which is half of the test piece at the hemisphere temperature.
Click here to see the Celignis Analysis Packages that determine Ash Melting Behaviour
Request a QuoteAsh Melting Behaviour
Major and Minor Elements in Foliage
We can also determine the levels of 13 different minor elements (such as arsenic, copper, and zinc) that may be present in Foliage.
Click here to see the Celignis Analysis Packages that determine Major and Minor Elements
Request a QuoteMajor and Minor Elements
Analysis of Foliage for Anaerobic Digestion
Biomethane potential (BMP) of Foliage
At Celignis we can provide you with crucial data on feedstock suitability for AD as well as on the composition of process residues. For example, we can determine the biomethane potential (BMP) of Foliage. The BMP can be considered to be the experimental theoretical maximum amount of methane produced from a feedstock. We moniotor the volume of biogas produced allowing for a cumulative plot over time, accessed via the Celignis Database. Our BMP packages also involve routine analysis of biogas composition (biomethane, carbon dioxide, hydrogen sulphide, ammonia, oxygen). We also provide detailed analysis of the digestate, the residue that remains after a sample has been digested. Our expertise in lignocellulosic analysis can allow for detailed insight regarding the fate of the different biogenic polymers during digestion.
Click here to see the Celignis Analysis Packages that determine BMP
Request a QuoteBMP
Physical Properties of Foliage
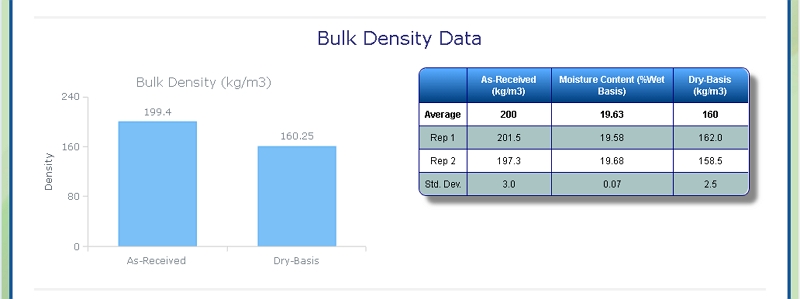
Bulk Density of Foliage
At Celignis we can determine the bulk density of biomass samples, including Foliage, according to ISO standard 17828 (2015). This method requires the biomass to be in an appropriate form (chips or powder) for density determination.
Click here to see the Celignis Analysis Packages that determine Bulk Density
Request a QuoteBulk Density
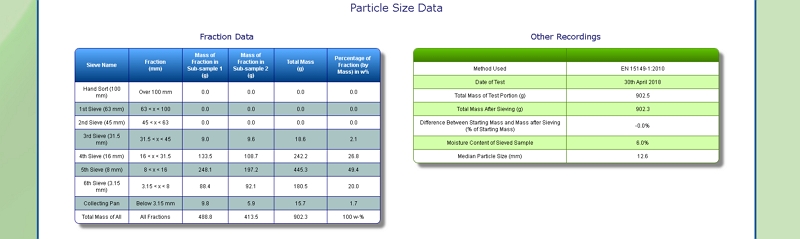
Particle Size of Foliage
Our lab is equipped with a Retsch AS 400 sieve shaker. It can accommodate sieves of up to 40 cm diameter, corresponding to a surface area of 1256 square centimetres. This allows us to determine the particle size distribution of a range of samples, including Foliage, by following European Standard methods EN 15149- 1:2010 and EN 15149-2:2010.
Click here to see the Celignis Analysis Packages that determine Particle Size
Request a QuoteParticle Size