Services for the Application and Evaluation of Enzymes
Background on Enzymatic Hydrolysis
Starch Biorefinery Enzymes
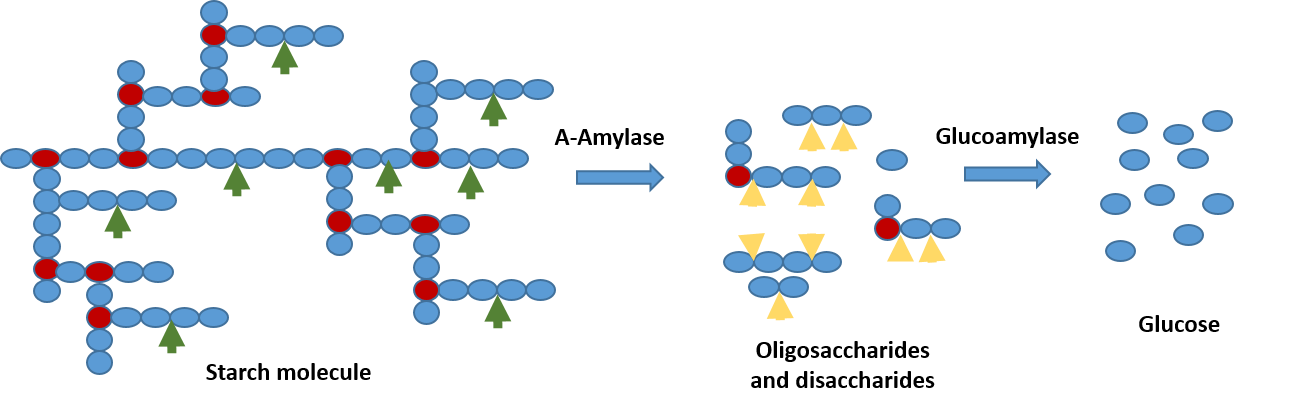
Alpha-amylase
Glucoamylase
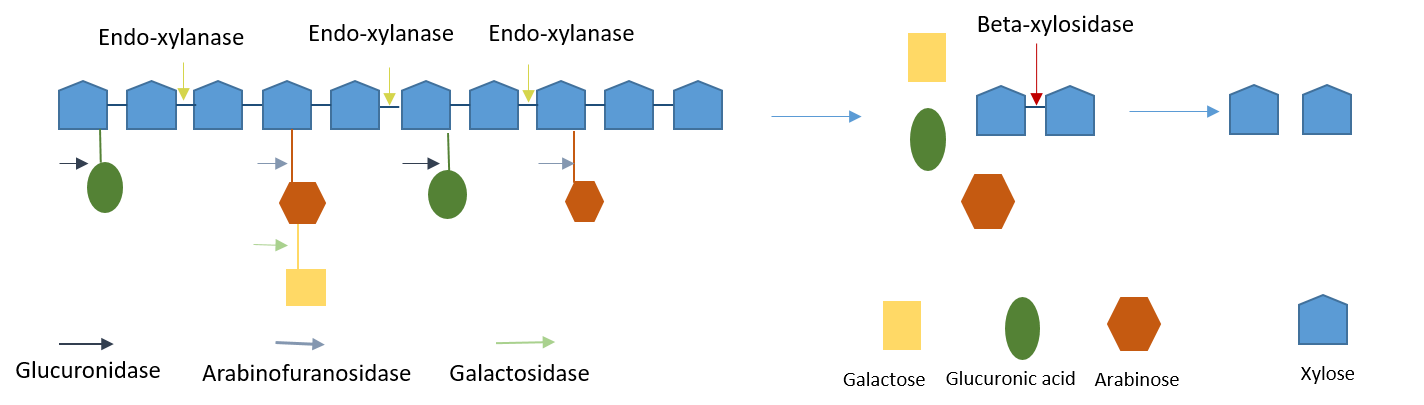
Lignocellulose Biorefinery Enzymes
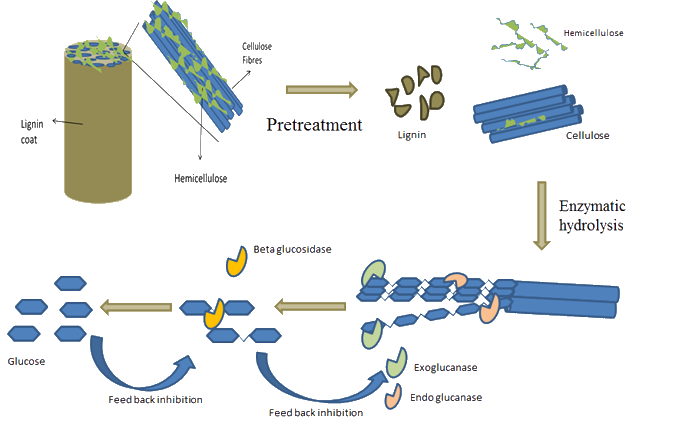
|
Enzymatic Hydrolysis Tests at Celignis
Evaluation of Biomass and Pre-Treated Biomass for Enzymatic Digestibility
These enzymatic hydrolysis tests can be carried out on native biomass and on pre-treated samples and we can either use a commercial enzyme mix or you can supply your own enzymes.
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Cellulose Conversion Yield, Xylan Conversion Yield, Combined Sugar Yield, Cellulose Conversion Rate, Xylan Conversion Rate
Evaluation of Pre-Treatment on Enzymatic Digestibility
As a result, several pre-treatment strategies to increase the accessibility of cellulose and enhance the digestibility of lignocellulosic biomass by enzymes have been developed. An effective pre-treatment will result in increased accessibility of cellulose to enzymes and thus in high concentrations, yields and productivities of sugars in the enzymatic hydrolysate when compared to the un-treated biomass.
At Celignis we can undertake the enzymatic hydrolysis of a pre-treated sample using analysis package P121 or we can undertake a more detailed study to evaluate the efficacy of pre-treatment. This study involves analysis package P121 being undertaken on the native (non-pretreated) sample with analysis package P122 used for the enzymatic hydrolysis of the pre-treated sample(s).
|
- Increase in Cellulose Accessibility after Pre-Treatment |
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Cellulose Conversion Yield, Xylan Conversion Yield, Combined Sugar Yield, Cellulose Conversion Rate, Xylan Conversion Rate, Increase in Cellulose Accessibility after Pre-Treatment, Percent Increase in Cellulose Conversion Efficiency, Percent Increase in Cellulose Conversion Rate
Based on their concentrations, these inhibitory compounds can negatively affect the fermentation process. For example, furans can inhibit cell replications at higher concentrations whilst weak acids can inhibit fermentation by accumulation of intracellular anions or by uncoupling of cell metabolism. Knowledge of the types of inhibitors in the enzyme hydrolysate and their concentrations will help in developing pre-treatment processes to avoid or reduce inhibitor formation and also to select suitable detoxification processes prior to fermentation.
Our cellulose saccharification test can be undertaken using pure cellulose or complex lignocellulosic substrates taken either from Celignis's repository or, alternatively, you can supply these substrates yourself.
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Cellulose Conversion Yield, Cellulose Conversion Rate
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Xylan Conversion Yield, Xylan Conversion Rate
|
|
|
|
Summary of Analysis Packages for Enzymatic Hydrolysis and Enzyme Activities
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Cellulose Conversion Yield, Xylan Conversion Yield, Combined Sugar Yield, Cellulose Conversion Rate, Xylan Conversion Rate
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Cellulose Conversion Yield, Xylan Conversion Yield, Combined Sugar Yield, Cellulose Conversion Rate, Xylan Conversion Rate, Increase in Cellulose Accessibility after Pre-Treatment, Percent Increase in Cellulose Conversion Efficiency, Percent Increase in Cellulose Conversion Rate
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Cellulose Conversion Yield, Cellulose Conversion Rate
Total Sugars in Enzyme Hydrolysate, Glucose in Enzyme Hydrolysate, Xylose in Enzyme Hydrolysate, Arabinose in Enzyme Hydrolysate, Mannose in Enzyme Hydrolysate, Galactose in Enzyme Hydrolysate, Rhamnose in Enzyme Hydrolysate, Cellobiose in Enzyme Hydrolysate, Enzymatic Hydrolysis Kinetics, Xylan Conversion Yield, Xylan Conversion Rate
Examples of Cellulosic Biomass Suitable for Enzymatic Hydrolysis Tests
This study evaluated the physicochemical properties and bioenergy potential of three distinct paper sludges: virgin pulp sludge (VP-PS), corrugated cardboard sludge (CR-PS), and tissue and printing paper sludge (TPR-PS). From the experimental runs, VP-PS exhibited the highest ethanol yield (46.8 ± 3.7 g L−1, 87.4% conversion), which can be attributed to its high glucan content and efficient enzymatic hydrolysis. Corrugated cardboard, despite its higher lignin content, demonstrated superior biogas and methane production (94.4 ± 8.3 L kg−1 and 54.4 ± 5.1 L CH4 kg−1 TSfed), likely due to its elevated xylan levels and favorable bulk density. The TPR-PS was characterized by high ash content and showed lower performance for both bioenergy production pathways but displayed improved solid handling due to its higher bulk density and lower water-holding capacity. These results provide a good explanation of the potential bioconversion pathways for the different paper sludge characteristics to maximize bioenergy yield. | ||
Brewers spent grains (BSGs) represent the largest quantity of solid waste from brewing, while xylooligosaccharides (XOS) produced from BSG show promising applications in food, beverage and health products. Production of XOS from a Weiss and malt BSG was scaled-up in steam explosion hydrothermal treatment using process conditions from bench-scale liquid hot water optimisations in stirred batch reactors. Three levels of moisture (15, 25 and 32% dry matter) achieved by screw press dewatering were evaluated by varying the treatment temperatures and times. Results show the highest XOS yields (73.1%) were obtained, for both BSGs, at process condition selected (180 C, 10 min) with 25% initial dry matter content. These yields were higher than reported bench-scale optimisations (61%), but obtained using 60% less water; hence, initial dry matter content was an important variable affecting XOS yield. The pilot-scale steam explosion results provide a departing point for a cost-effective commercial production of XOS from BSG. | |
A techno-economic assessment (TEA) of enzymatic hydrolyses of a municipal solid waste (MSW)-derived pulp was performed to compare various bioprocessing configurations for the production of platform sugars at both pilot and demonstration scales (two-stage continuous, batch, and two-stage fed-batch). The configurations modeled used either rotary drum and/or continuous stirred tank reactors. By using reaction kinetics and public vendor's quotes, economic analyses were calculated for each of the proposed systems: capital expenditure (CapEx); operation expenditure (OpEx); revenue and profit; return on investment (ROI); and payback period (PP). The TEA showed that a two-stage continuous configuration with a total residence time of 54 h (6 and 48 h for primary and secondary stages) was the best option for obtaining sugars, showing sevenfold higher enzyme productivity and better profit than the reference systems. Although pilot-scale enzymatic hydrolysis demonstrated an unprofitable process, this was mainly due to the high associated enzyme cost. Increasing the scale diminished this problem, leading to higher profit per processed unit (GBP/kg lignocellulosic sugars). From an investment perspective, the two-stage 6/48 configuration gave a more attractive ROI and PP than the other designs. | |
weet sorghum bagasse displays many characteristics rendering it a promising substrate for lignocellulosic ethanol production. In this study, the steam pretreatment catalyst, enzymatic hydrolysis and the substrate loading for the fermentation were investigated in order to maximise the production of ethanol from the feedstock. The results deemed water as a sufficient pretreatment catalyst since the SO2 impregnation of the biomass did not produce any significant beneficial effects on the yield of ethanol produced. The preferred pretreatment and enzymatic hydrolysis conditions were incorporated in a fed-batch simultaneous saccharification and fermentation (SSF) process using pressed-only (not washed) WIS at a final solid loading of 13% (w/w) that resulted in the targeted ethanol concentration of 39 g/L with a corresponding yield of 82% of the theoretical maximum. Yeast inhibition coupled with significant glucose accumulation was observed at higher solid loadings of 16% and 20%. Ultimately, the sweet sorghum bagasse could be integrated into existing ethanol production regimes to improve the global bioenergy production. | |
Commercial production of inulooligosaccharides (IOS) relies largely on chicory roots. However, Jerusalem artichoke (JA) tubers provide a suitable alternative due to their high inulin content and low cultivation requirements. In this study, three inulin-rich substrate preparations from JA were investigated to maximize IOS production, namely powder from dried JA tuber slices (Substrate 1), solid residues after extracting protein from the JA powder (Substrate 2) and an inulin-rich fraction extracted from protein extraction residues (Substrate 3). The preferred temperature, pH and inulin substrate concentration were determined after which enzyme dosage and extraction time were optimized to maximize IOS extraction from the three substrates, using pure chicory inulin as benchmark. Under the optimal conditions, Substrate 3 resulted in the highest IOS yield of 82.3% (w/winulin). However, IOS production from the Substrate 1 proved more efficient since it renders the highest overall IOS yield (mass of IOS per mass of the starting biomass). In the case of co-production of protein and IOS from the JA tuber in a biorefinery concept, IOS production from the Substrate 2 is preferred since it reduces the inulin losses incurred during substrate preparation. For all the inulin-rich substrates studied, an enzyme dosage of 14.8 U/ginulin was found to be optimal at reaction time less than 6 h. JA tuber exhibited excellent potential for commercial production of IOS with improved yield and the possible advantage of a reduced biomass cost. | |
BACKGROUND | |
Brewers' spent grains (BSG) make up to 85% of a brewery's solid waste, and is either sent to landfill or sold as cheap animal feed supplement. Xylo-oligosaccharides (XOS) obtained from BSG are antioxidants and prebiotics that can be used in food formulations as low-calorie sweeteners and texturisers. The effect of extremely low acid (ELA) catalysis in liquid hot water (LHW) hydrothermal treatment (HTT) was assessed using BSG with dry matter contents of 15% and 25%, achieved by dewatering using a screw press. Batch experiments at low acid loadings of 5, 12.5 and 20 mg/g dry mass and temperatures of 120, 150 and 170 C significantly affected XOS yield at both levels of dry mass considered. Maximum XOS yields of 76.4% (16.6 g/l) and 65.5% (31.7 g/l) were achieved from raw BSG and screw pressed BSG respectively, both at 170 C and using 5 mg acid/g dry mass, after 15 min and 5 min, respectively. These XOS yields were obtained with BSG containing up to 63% less water and temperatures more than 20 C lower than that reported previously. The finding confirms that ELA dosing in LHW HTT allows lowering of the required temperature that can result in a reduction of degradation products, which is especially relevant under high solid conditions. This substantial XOS production intensification through higher solid loadings in HTT not only achieved high product yield, but also provided benefits such as increased product concentrations and decreased process heat requirements. | |
Short-chain fructooligosaccharides (scFOS) are nutraceuticals with numerous applications in the food and pharmaceutical industries. The production of scFOS using immobilized biocatalysts offers some functional and technical advantages over free enzyme counterparts. To investigate the economic potential of the immobilized enzyme system relative to the free enzyme system, a techno-economic comparison was conducted on three methods of scFOS production (powder and syrup forms) at a capacity of 2000?t per annum (tpa) by enzymatic synthesis from sucrose: the free enzyme (FE), calcium alginate immobilized enzyme (CAIE), and amberlite IRA 900 immobilized enzyme (AIE) systems. These processes were simulated in Aspen Plus to obtain the mass and energy balances and to estimate the operating and capital costs, followed by economic evaluation and sensitivity analysis. Profitability analysis showed that all three systems are economically viable as their associated minimum selling prices (MSP) were well below the scFOS market price of 5 $ kg-1. However, the FE system was the most profitable with the lowest MSP of 2.61 $ kg-1 because the savings on cost as a result of enzyme immobilization could not offset the additional costs associated with immobilization. Sensitivity analysis demonstrated that total operating cost, fixed capital investment, and internal rate of return (% IRR) have the greatest effects on the MSP. Furthermore, the syrup form of scFOS production leads to 29% less MSP, compared to powder form. In addition, the studied plant capacities of 5000 and 1000?tpa showed 10% and 16% reductions on MSP respectively. | |
Jerusalem artichoke (JA) is a crop with excellent potential for application in biorefineries. It can resist drought, pests, and diseases and can thrive well in marginal lands with little fertilizer application. The JA tubers contain considerable quantities of inulin, which is suitable for the production of inulooligosaccharides (IOS), as a high-value prebiotic, dietary fiber. In this study, five JA tuber biorefinery scenarios were simulated in Aspen Plus and further evaluated by techno-economic and sensitivity analyses. Production of IOS, proteins and animal feed was studied in scenarios A and C, applying various biorefinery configurations. Scenario B explored the option of producing only IOS and the sale of residues as animal feed. Scenarios D and E investigated the economic potential of biofuel generation from residues after IOS and protein production by generation of biogas and ethanol respectively, from residues. Based on the chosen economic indicators, scenario B resulted in the lowest minimum selling price (MSP) of 3.91 US$ kg-1 (market price 5.0 US$ kg-1) with correspondingly reduced total capital investment (TCI) and total operating cost (TOC) per mass unit produced of IOS of 18.91 and 2.59 US$ kg-1 respectively, compared with other studied scenarios. Considering the set production scale, it is more profitable when the residues are sold as animal feed instead of being converted into biofuel, due to the capital-intensive nature of the biofuel production processes. The coproduction of protein had a negative impact on the economics of the process as the associated capital and operating expenditure outweighed the associated revenue. | |
The immobilization of B-fructofuranosidase for short-chain fructooligosaccharide (scFOS) synthesis holds the potential for a more efficient use of the biocatalyst. However, the choice of carrier and immobilization technique is a key to achieving that efficiency. In this study, calcium alginate (CA), Amberlite IRA 900 (AI900) and Dowex Marathon MSA (DMM) were tested as supports for immobilizing a novel engineered B-fructofuranosidase from Aspergillus japonicus for scFOS synthesis. Several immobilization parameters were estimated to ascertain the effectiveness of the carriers in immobilizing the enzyme. The performance of the immobilized biocatalysts are compared in terms of the yield of scFOS produced and reusability. The selection of carriers and reagents was motivated by the need to ensure safety of application in the production of food-grade products. The CA and AI900 both recorded impressive immobilization yields of 82 and 62%, respectively, while the DMM recorded 47%. Enzyme immobilizations on CA, AI900 and DMM showed activity recoveries of 23, 27, and 17%, respectively. The CA, AI900 immobilized and the free enzymes recorded their highest scFOS yields of 59, 53, and 61%, respectively. The AI900 immobilized enzyme produced a consistent scFOS yield and composition for 12 batch cycles but for the CA immobilized enzyme, only 6 batch cycles gave a consistent scFOS yield. In its first record of application in scFOS production, the AI900 anion exchange resin exhibited potential as an adequate carrier for industrial application with possible savings on cost of immobilization and reduced technical difficulty. | |
Disposal of waste sludges produced in large amounts in the pulp and paper industry imposes significant environmental and economical problems. One strategy to address these issues involves revalorization of paper mill sludges by their application as substrates for microbial production of biotechnologically relevant enzymes. The application of lignocellulolytic enzymes in paper, textile and bioenergy industries is encouraged in order to decrease chemicals and energy consumptions. In the following work, deinking sludge was assessed as a substrate for production of lignocellulases. Based on the results of growth and activity screenings, Pleurotus ostreatus PLAB was chosen as the most promising candidate among 30 tested strains and its secretome was further studied by quantitative enzyme assays and mass spectrometry. While endoglucanase and xylanase activities detected in P. ostreatus secretome produced on deinking sludge were similar to activities of cultures grown on other lignocellulosic substrates, average laccase activity was significantly higher (46?000 U/kg DIS). Mass spectrometry identification of the most prominent proteins in the secretome of the target strain confirmed that significant amounts of different lignin-modifying oxidases were produced on this substrate despite its low lignin content, indicating the presence of other inducible compounds. The findings of this study suggest deinking sludge may represent a good substrate for fungal production of the aforementioned enzymes with broad biotechnological applications, including bioremediation, paper and bioenergy industries. | |
Biobutanol has gained attention as an alternative renewable transportation fuel for its superior fuel properties and widespread applications in chemical industry, primarily as a solvent. Conventional butanol fermentation has drawbacks that include strain degeneration, end-product toxicity, by-product formation, low butanol concentrations and high substrate cost. The complexity of Clostridium physiology and close control between sporulation phase and ABE fermentation has made it demanding to develop industrially potent strains. In addition to the isolation and engineering of superior butanol producing bacteria, the development of advanced cost-effective technologies for butanol production from feedstock like lignocellulosic biomass has become the primary research focus. High process costs associated with complex feedstocks, product toxicity and low product concentrations are few of the several bioprocess challenges involved in biobutanol production. The article aims to assess the challenges in lignocellulosic biomass to biobutanol conversion and identify key process improvements that can make biobutanol commercially viable. | |
The paper and pulp industry is one of the major industries that generate large amount of solid waste with high moisture content. Numerous opportunities exist for valorisation of waste paper sludge, although this review focuses on primary sludge with high cellulose content. The most mature options for paper sludge valorisation are fermentation, anaerobic digestion and pyrolysis. In this review, biochemical and thermal processes are considered individually and also as integrated biorefinery. The objective of integrated biorefinery is to reduce or avoid paper sludge disposal by landfilling, water reclamation and value addition. Assessment of selected processes for biorefinery varies from a detailed analysis of a single process to high level optimisation and integration of the processes, which allow the initial assessment and comparison of technologies. This data can be used to provide key stakeholders with a roadmap of technologies that can generate economic benefits, and reduce carbon wastage and pollution load. | ||
Paper sludge (PS) from the paper and pulp industry consists primarily of cellulose and ash and has significant potential for ethanol production. Thirty-seven PS samples from 11 South African paper and pulp mills exhibited large variation in chemical composition and resulting ethanol production. Simultaneous saccharification and fermentation (SSF) of PS in fed-batch culture was investigated at high solid loadings and low enzyme dosages. Water holding capacity and viscosity of the PS influenced ethanol production at elevated solid loadings of PS. High viscosity of PS from virgin pulp mills restricted the solid loading to 18% (w/w) at an enzyme dosage of 20 FPU/gram dry PS (gdPS), whereas an optimal solid loading of 27% (w/w) was achieved with corrugated recycle mill PS at 11 FPU/gdPS. Ethanol concentration and yield of virgin pulp and corrugated recycle PS were 34.2 g/L at 66.9% and 45.5 g/L at 78.2%, respectively. | |
Next-generation biofuels from renewable sources have gained interest among research investigators, industrialists, and governments due to major concerns on the volatility of oil prices, climate change, and depletion of oil reserves. Biobutanol has drawn signicant attention as an alternative transportation fuel due to its superior fuel properties over ethanol. e advantages of butanol are its high energy content, better blending with gasoline, less hydroscopic nature, lower volatility, direct use in convention engines, low corrosiveness, etc. Butanol production through (acetone, butanol, and ethanol) ABE fermentation is a well-established process, but it has several drawbacks like feedstock cost, strain degeneration, product toxicity, and low product concentrations. Lignocellulosic biomass is considered as the most abundant, renewable, low-cost feedstock for biofuels. Production of butanol from lignocellulosic biomass is more complicated due to the recalcitrance of feedstock and inhibitors generated during the pretreatment and hydrolysis process. Advanced fermentation and product recovery techniques are being researched to make biobutanol industrially viable. | |
Bioethanol and biobutanol hold great promise as alternative biofuels, especially for transport sector, because they can be produced from lignocellulosic agro-industrial residues. From techno-economic point of view, the bioprocess for biofuels production should involve minimal processing steps. Consolidated bioprocessing (CBP), which combines various processing steps such as pretreatment, hydrolysis and fermentation in a single bioreactor, could be of great relevance for the production of bioethanol and biobutanol or solvents (acetone, butanol, ethanol), employing clostridia. For CBP, Clostridium holds best promise because it possesses multi-enzyme system involving cellulosome and xylanosome, which comprise several enzymes such as cellulases and xylanases. The aim of this article was to review the recent developments on enzyme systems of clostridia, especially xylanase and cellulase with an effort to analyse the information available on molecular approaches for the improvement of strains with ultimate aim to improve the efficiencies of hydrolysis and fermentation. | |
Biobutanol from lignocellulosic biomass has gained much attention due to several advantages over bioethanol. Though microbial production of butanol through ABE fermentation is an established technology, the use of lignocellulosic biomass as feedstock presents several challenges. In the present study, biobutanol production from enzymatic hydrolysate of acid pretreated rice straw was evaluated using Clostridium sporogenes BE01. This strain gave a butanol yield of 3.43 g/l and a total solvent yield of 5.32 g/l in rice straw hydrolysate supplemented with calcium carbonate and yeast extract. Hydrolysate was analyzed for the level of inhibitors such as acetic acid, formic acid and furfurals which affect the growth of the organism and in turn ABE fermentation. Methods for preconditioning the hydrolysate to remove toxic end products were done so as to improve the fermentation efficiency. Conditions of ABE fermentation were fine tuned resulting in an enhanced biobutanol reaching 5.52 g/l. | |
Aspergillus niger NII-08121/MTCC 7956 exhibited differences in expression of ?-glucosidase (BGL) in response to carbon sources provided in the medium. Activity staining with methyl umbelliferyl ?-d-glucopyranoside (MUG) indicated that four different isoforms of BGL were expressed when A. niger was grown under submerged fermentation with either lactose or cellulose, whereas only two were expressed when wheat bran or rice straw was used as the carbon source. Among the four isoforms of BGL expressed during lactose supplementation, two were found to retain 92% and 82% activity respectively in presence of 250 mM glucose in the MUG assay. The major ?-glucosidase (BGL1) was purified to homogeneity by electro elution from a Native PAGE gel. The purified 120 kDa protein was active at 50 �C and was stable for 48 h without any loss of activity. The optimum pH and temperature were 4.8 and 70 �C respectively. | |
Rice straw is an attractive lignocellulosic material for bioethanol production since it is one of the most abundant renewable resources. It has several characteristics, such as high cellulose and hemicelluloses content that can be readily hydrolyzed into fermentable sugars. But there occur several challenges and limitations in the process of converting rice straw to ethanol. The presence of high ash and silica content in rice straw makes it an inferior feedstock for ethanol production. One of the major challenges in developing technology for bioethanol production from rice straw is selection of an appropriate pretreatment technique. The choice of pretreatment methods plays an important role to increase the efficiency of enzymatic saccharification thereby making the whole process economically viable. The present review discusses the available technologies for bioethanol production using rice straw. | |
Biomass feedstock having less competition with food crops are desirable for bio-ethanol production and such resources may not be localized geographically. A distributed production strategy is therefore more suitable for feedstock like water hyacinth with a decentralized availability. In this study, we have demonstrated the suitability of this feedstock for production of fermentable sugars using cellulases produced on site. Testing of acid and alkali pretreatment methods indicated that alkali pretreatment was more efficient in making the sample susceptible to enzyme hydrolysis. Cellulase and ?-glucosidase loading and the effect of surfactants were studied and optimized to improve saccharification. Redesigning of enzyme blends resulted in an improvement of saccharification from 57% to 71%. A crude trial on fermentation of the enzymatic hydrolysate using the common baker�s yeast Saccharomyces cerevisiae yielded an ethanol concentration of 4.4 g/L. | |





