Analysis Packages for Bioenergy Parameters
Our laboratory is equipped with a number of state-of-the art items of equipment that allow us to determine the most important combustion-related properties of your biomass including calorific value, volatile matter, elemental composition, moisture content, ash composition and ash melting behaviour.
Moisture and ash contents are of crucial importance for combustion. This is reflected in our online, Excel, and pdf reports where all data for bioenergy-related parameters are expressed on dry-mass, as-received, and dry-ash free bases, according to standard method EN 15296:2011.
Water is generally held in biomass in two ways - either as a free liquid and vapour that is contained in the cell cavities, or as a molecule that is bound within the cell walls.
Moisture content tends to vary widely with biomass species, age, geographic locations and genetic differences. It also varies between different anatomical fractions of the same plant and throughout the year.
Similarly, our data regarding the biomethane potential of anaerobic digestion feedstocks are reported on as-received as well as dry bases.
Analysis Packages for Moisture Content
Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing), Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium
Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Specific Surface Area (Nitrogen Gas Adsorption), Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Ash content can vary greatly between plant species, and is generally higher in agricultural residues. The ash present in plants will depend on their stage of growth, the time of year, and their location. The leaching of stored biomass may reduce the level of inorganics in some instances.
At Celignis with our elemental analysis methods we are able to characerise a wide range of major and minor analytes present in biomass ash. Click here to see our relevant analysis packages.
Analysis Packages for Ash Content
Total Sugars, Glucose, Xylose, Mannose, Arabinose, Galactose, Rhamnose, Lignin (Klason), Lignin (Acid Soluble), Acid Insoluble Residue, Extractives (Ethanol-Soluble), Extractives (Water-Soluble), Extractives (Exhaustive - Water then Ethanol), Extractives (Water-Insoluble, Ethanol Soluble) , Ash, Ash (Acid Insoluble)
Total Sugars, Glucose, Xylose, Mannose, Arabinose, Galactose, Rhamnose, Lignin (Klason), Lignin (Klason - Protein Corrected), Lignin (Acid Soluble), Acid Insoluble Residue, Extractives (Ethanol-Soluble), Extractives (Water-Soluble), Extractives (Exhaustive - Water then Ethanol), Extractives (Water-Insoluble, Ethanol Soluble) , Ash, Ash (Acid Insoluble), Glucuronic Acid, Galacturonic Acid, 4-O-Methyl-D-Glucuronic Acid, Protein Content of Acid Insoluble Residue, Carbon Content of Acid Insoluble Residue, Hydrogen Content of Acid Insoluble Residue, Nitrogen Content of Acid Insoluble Residue, Sulphur Content of Acid Insoluble Residue, Xylitol, Sucrose, Fructose, Sorbitol, Trehalose
Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing), Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium
Total Sugars, Glucose, Xylose, Mannose, Arabinose, Galactose, Rhamnose, Lignin (Klason), Lignin (Acid Soluble), Carbon, Extractives (Ethanol-Soluble), Extractives (Water-Soluble), Extractives (Exhaustive - Water then Ethanol), Extractives (Water-Insoluble, Ethanol Soluble) , Ash, Ash (Acid Insoluble), Starch, Pectin, Glucuronic Acid, Galacturonic Acid, 4-O-Methyl-D-Glucuronic Acid
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Analysis Packages for Ash Composition
Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing), Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium
Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Specific Surface Area (Nitrogen Gas Adsorption), Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
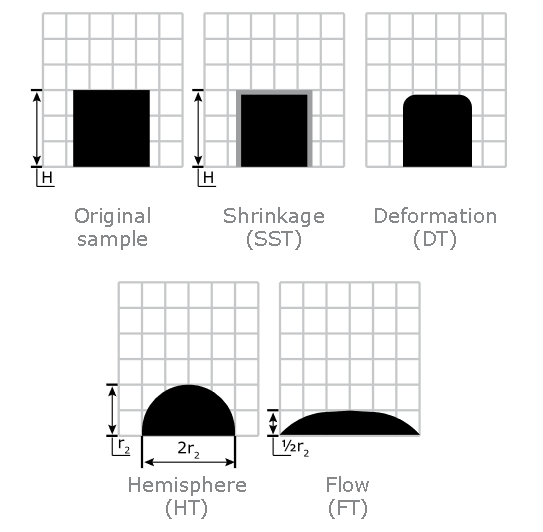
Ash Shrinkage Starting Temperature (SST) - This occurs when the area of the test piece of biomass ash falls below 95% of the original test piece area.
Ash Deformation Temperature (DT) - The temperature at which the first signs of rounding of the edges of the test piece occurs due to melting.
Ash Hemisphere Temperature (HT) - When the test piece of biomass ash forms a hemisphere (i.e. the height becomes equal to half the base diameter).
Ash Flow Temperature (FT) - The temperature at which the biomass ash is spread out over the supporting tile in a layer, the height of which is half of the test piece at the hemisphere temperature.
Analysis Packages for Ash Melting Behaviour
Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing), Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium
Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Specific Surface Area (Nitrogen Gas Adsorption), Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Lignocellulosic biomass is mainly composed of carbohydrates (in the form of the polysaccharides cellulose and hemicellulose) and lignin. Carbohydrates, having an elemental composition of (CH2O)n, will have a generally uniform carbon content of 40%, which is lower than many other biomass mass constituents such as lignin which has an average carbon content of approximately 60-65%.
Typically carbon, hydrogen, nitrogen, and sulphur can be determined in the same analytical method and the oxygen content can then be calculated as the remaining mass after the ash content has been considered.
We use an Elementar MACRO Cube unit for the ultimate analysis of samples. It allows for large sample sizes (e.g. around 40mg) to be used than many other elemental analysers, allowing us to improve the precision in the analysis of biomass and waste samples.
Analysis Packages for Ultimate Composition
Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing), Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium
Total Sugars, Glucose, Xylose, Mannose, Arabinose, Galactose, Rhamnose, Lignin (Klason), Lignin (Acid Soluble), Carbon, Extractives (Ethanol-Soluble), Extractives (Water-Soluble), Extractives (Exhaustive - Water then Ethanol), Extractives (Water-Insoluble, Ethanol Soluble) , Ash, Ash (Acid Insoluble), Starch, Pectin, Glucuronic Acid, Galacturonic Acid, 4-O-Methyl-D-Glucuronic Acid
Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Specific Surface Area (Nitrogen Gas Adsorption), Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
There are several units of measurement. The caloric, or higher heating value (HHV), is independent of moisture content and reliant on the chemical composition of the material. A linear relationship exists between the heat of combustion and the carbon content of the substrate while oxygen, nitrogen and inorganic elements tend to reduce the value.
The Lower Heating Value (LHV), or effective heating value, is perhaps more relevant than the HHV in practical operations. It considers not only the energy required to vaporise the moisture of the biomass but also that necessary to vaporise the water generated when the hydrogen and oxygen elements of the biomass combine. Hydrogen content then becomes a reducing factor in the heating value.
At Celignis we use a Parr 6200 bomb calorimeter to directly determine the gross calorific value (higher heating value) of samples. The data from ultimate analysis are then used to calculate the net calorific value (lower heating value).
Analysis Packages for Calorific Value
Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing), Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium
Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Aluminium, Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
Thernogram - Under Nitrogen, Thermogram - Under Air, Moisture, Inherent Moisture, Ash Content (815C), Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Organic Carbon, Inorganic Carbon, Chlorine, Volatile Matter, Fixed Carbon, Specific Surface Area (Nitrogen Gas Adsorption), Calcium, Iron, Magnesium, Phosphorus, Potassium, Silicon, Sodium, Titanium, Gross Calorific Value, Net Calorific Value, Ash Shrinkage Starting Temperature (Reducing), Ash Deformation Temperature (Reducing), Ash Hemisphere Temperature (Reducing), Ash Flow Temperature (Reducing)
The biofuel production potentials for encroacher and invasive bush biomass species found in Southern Africa were assessed using different valorization routes. Theoretical models were employed to calculate the biofuel yields. The gasification-catalytic route produced highest ethanol yields (450–488 L/t) while the lowest values were from enzymatic/acid hydrolysis-to-fermentation route. Blue gum gave the highest ethanol yields. Biodiesel and naphtha yields produced through Fischer-Tropsch synthesis were highest for blue gum (196 L/t) and lowest for Acacia raficiens (176 L/t). The highest biogas and biomethane potential of 458 L/kg.VS and 229 L/kg.VS respectively were obtained from black wattle while the respective lower values (270 L/kg.VS and 132 L/kg.VS) were recorded for blue gum. Senegalia mellifera gave the highest torrefied biofuel energy and mass yields at 0.92 and 0.97 respectively while black wattle had the lowest mass and energy yields at 0.75 and 0.83 respectively. From an energy yield basis, the acid hydrolysis-fermentation route yielded an average of 3.69 GJ/t of biomass while the highest yields came from the gasification-catalytic conversion route which was 9.7 GJ/t. The average energy yield variations across biomass species ranged 5.11–6.19 GJ/t which is around 30 % of the raw biomass' calorific value. These early results provide insights towards the best pairing of appropriate biomass species and energy conversion route. Further evaluations of these biomass-valorization technology pairing to unpack process efficiencies, cost and kinetics are required using real process experiments instead of using theoretical models. These additional tests should include sustainability assessment to guide future commercialization decisions. | |