Separate Hydrolysis and Fermentation (SHF) of Lignocellulosic Biomass
Lignocellulose Hydrolysis
Lignocellulosic biomass, primarily comprised of cellulose, hemicellulose, and lignin, is an abundant and renewable resource that holds great promise as a source of biofuels and renewable biobased chemicals and biomaterials. Lignocellulosic biomass can be processed in a number of ways, one is through the hydrolysis of the structural polysaccharides (cellulose and hemicellulose) into their constituent sugars, a reaction commonly facilitated by acid or enzymes, followed by the fermentation of these sugars by yeast or other microorganisms.Click below to read more about the hydrolysis of lignocellulosic biomass.
Get more info...Biomass Hydrolysis
Enzymatic Hydrolysis
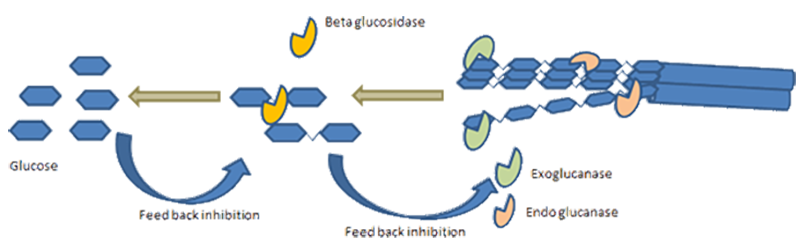
In enzymatic hydrolysis cellulases and hemicellulases play a critical role, working synergistically to cleave the glycosidic linkages in cellulose and hemicellulose, respectively. However, depending on the type of pretreatment process involved, hydrolysis of hemicellulose may not be necessary, since it may have already taken place in the pretreatment leading to the hemicellulose sugars being in the liquid output of the pretreatment with the solid residue mostly containing cellulose (plus lignin, again dependent on the type of pretreatment).The enzymatic hydrolysis step can be carried out under relatively mild conditions, which makes it a more energy-efficient and environmentally friendly approach compared to chemical hydrolysis methods.
SHF Process and its Advantages
SHF is characterized by two distinct stages: the hydrolysis of cellulose (and hemicellulose, if present after the pretreatment) into monomeric sugars, followed by the fermentation of these sugars into biofuels.Some of the advantages of the Separate Hydrolysis and Fermentation (SHF) approach are described below:
- Optimization of Individual Stages - The conditions that favour enzymatic hydrolysis (e.g. temperature, pH, and enzyme concentrations) do not necessarily coincide with those that are best for fermentation. In SHF, each process can be performed under its own optimal conditions. For instance, the hydrolysis stage can be carried out at a higher temperature, which is typically favorable for the enzymes but not for the fermenting microorganisms. This independent optimisation often results in higher yields of fermentable sugars from hydrolysis and better product yields from fermentation.
- Use of Well-Established Industrial Yeasts - SHF allows for the use of traditional, well-established yeast strains in the fermentation stage, at least when the fermentation of pentose sugars is not required (for example when the hemicellulose is removed in the pretreatment stage). These yeasts, such as Saccharomyces cerevisiae, are known for their robustness and high ethanol tolerance, which can result in high ethanol yields (where ethanol is the target of fermentation). These strains are also more resistant to inhibitors that might be present in the hydrolysate.
- Flexibility in Process Design and Operation - Due to the separate nature of the stages, different technologies and reactors can be used for the hydrolysis and fermentation steps, offering greater flexibility in process design. For instance, hydrolysis can be performed in a high-shear reactor to promote enzymatic activity, while fermentation can be conducted in a traditional stirred-tank fermenter.
Disadvantages of SHF
- Longer Processing Times - Since hydrolysis and fermentation occur separately, the total process time for SHF tends to be longer compared to simultaneous processes. This not only increases operational costs, but it also necessitates the use of holding tanks for storing the intermediate products, thus escalating the capital costs and infrastructure requirements.
- Product Inhibition During Hydrolysis - In the SHF process, the hydrolysis and fermentation stages are not simultaneous, which means that the sugars produced during hydrolysis do not immediately undergo fermentation. This means that the process is more likely to be affected by the inhibitory effect that high sugar concentrations can have on the enzymes involved in hydrolysis. This may result in the incomplete hydrolysis of cellulose and hemicellulose.
- Potential for Sugar Degradation - During the hydrolysis step, the produced sugars can degrade if they remain in the reactor for extended periods under acidic or high-temperature conditions, resulting in reduced product yields.
Simultaneous Saccharification and Fermentation (SSF)
Simultaneous Saccharification and Fermentation (SSF) is a process where enzymatic hydrolysis and fermentation occur concurrently in the same reactor.Advantages of SHF over SSF:
- Independent Optimization - The separate nature of the hydrolysis and fermentation stages allows each process to be optimized independently for parameters such as temperature, pH, and enzyme concentration. In contrast, in SSF processes it can be challenging to find a single set of conditions that are optimal for both enzymatic hydrolysis and fermentation. For example, the ideal temperatures for these processes usually differ.
- Use of Well-Established Microorganisms - SHF allows for the use of traditional, robust yeast strains in the fermentation stage, which can lead to high product yields. In contrast, more novel, less-robust and less well established microorganisms tend to be used in SSF.
- Flexibility in Process Design and Operation - Different technologies and reactors can be used for the hydrolysis and fermentation steps, offering greater flexibility in process design.
- Longer Processing Times - Because hydrolysis and fermentation occur separately, the total process time tends to be longer than SSF.
- Increased Risk of Contamination - The longer duration of the process and multiple stages increase the risk of microbial contamination. In contrast, in SSF the shorter process time and single stage reduce the opportunities for contamination.
- Potential for Sugar Degradation - Sugars produced in hydrolysis can degrade if they remain in the reactor for extended periods.
- Possible Product Inhibition - The accumulation of sugars during the hydrolysis phase can potentially inhibit enzyme activity. Whilst in SSF, since the sugars are fermented as soon as they are produced during hydrolysis, there is less risk of high sugar concentrations inhibiting enzyme activity.
Get more info...SSF
Consolidated Bioprocessing (CBP)
Consolidated Bioprocessing (CBP) is a more integrated approach in which the enzyme production, hydrolysis, and fermentation steps all occur in one step and one reactor. CBP has the potential to significantly reduce costs and simplify the biofuel production process. However, the implementation of CBP is currently challenging because it requires a single microorganism or a consortium of microorganisms that can efficiently perform all the required functions.Advantages of SHF over CBP:
- Independent Optimization - The separate nature of the hydrolysis and fermentation stages allows each process to be optimized independently.
- Use of Well-Established Microorganisms - SHF allows for the use of traditional, robust yeast strains in the fermentation stage, which can lead to high product yields.
- Flexibility in Process Design and Operation - Different technologies and reactors can be used for the hydrolysis and fermentation steps.
- More-Established Technology - The development of CBP technologies is still at the R&D stage whilst SHF technologies have been deployed at the commercial scale.
- Longer Processing Times - The total process time for SHF tends to be longer compared to CBP.
- Increased Risk of Contamination - The longer duration of the process increases the risk of microbial contamination.
- Potential for Sugar Degradation - The sugars produced during hydrolysis can degrade if they remain in the reactor for extended periods.
- Cost of Enzymes - In CBP the enzymes are produced directly by the microorganisms inside the reactor. In contrast, SHF processes require that enzymes are added at the hydrolysis stage. Either these SHF enzymes have to be purchased, adding significantly to OPEX, or they need to be manufactured on-site, adding to OPEX and CAPEX.
Get more info...CBP
1. Understanding Your Requirements
Prior to undertaking bioprocess projects we learn from our clients what their targets are from the process as well as whether there are any restrictions or requirements that may need to form the boundaries of the work that we undertake. These help to guide us to then prepare a potential bioprocess development project.
2. Detailed Feedstock Analysis
In cases where you have already selected a feedstock for the bioprocess, we would then undertake a detailed compositional analysis (P10 or, ideally, P19) of representative samples of that feedstock.
In cases where the feedstock has not yet been selected we can review your list of candidate feedstocks, selecting top candidates based on our prior experience in their analysis and bioprocessing. If you do not have a list of candidate feedstocks then we can provide one, based on your location and the requirements outlined in Stage 1. We would then analyse in detail these priority feedstocks and come to a decision, based on the compositional data and other relevant factors (e.g. price, supply, consistency etc.) on a selected feedstock for the project.
At this point of the project, the Celignis Bioprocess team typically meet to discuss and prepare a project proposal for the development of a SHF bioprocess from this feedstock. After this proposal is reviewed by the client, and revised if needed, we are then ready to start work on the next stages.
3. Pretreatment (Lab-Scale)
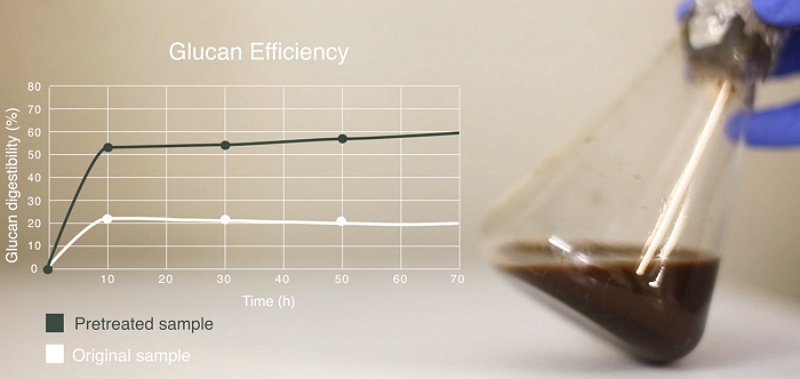
If our project involves the SHF-processing of a virgin biomass feedstock then it is likely that a pretreatment stage will be necessary in order to provide a suitable substrate for subsequent hydrolysis and fermentation. Hence, this stage of the project will involve undertaking a number of pretreatment experiments, covering a variety of process conditions. We follow a scientifically-based Design of Experiments (DoE) protocol where the criteria and boundaries for this DoE are formulated in close collaboration with our clients, considering the chemistry of the feedstock(s) and our understandings of the mechanisms of biomass pretreatment.
We usually recommend that these initial optimisation experiments are undertaken at the lab-scale (around TRL3) in order to reduce costs and the length of the project. For each experiment we analyse the solid and liquid outputs of the pretreatment process, leading to a detailed data-set where effects of process conditions on the yield and composition of the various streams can be explored and mapped.
We can also undertake a second iteration of lab-scale experiments in order to fine-tune the conditions based on the knowledge gained in the initial experiments.
4. SHF Stage 1 (Hydrolysis) Optimisation
At this stage the focus is on optimising the enzymatic hydrolysis of the pretreated biomass. This will involve evaluating the effects of varying a number of different process conditions (e.g. solid-loading, enzyme-loading, enzyme-type, temperature, reaction times, mixing). Hence, this stage of the project involves a DoE being undertaken (like in Stage 3) where these conditions are narrowed down. Again, these optimisation experiments are usually undertaken at the lab-scale in order to accelerate the outputs and reduce project costs.
This stage usually follows the pretreatment optimisation activities, however it is possible that there can be some overlap in order to reach the final project outputs more quickly.
5. SHF Stage 2 (Fermentation) Optimisation
Here we work on optimising the fermentation step of the process so that the target product(s) can be obtained sustainably and in high yields. As with Stage 4 (Hydrolysis Optimisation), this work will involve a series of experiments (DoE) focused on finding the best combination of process parameters. Due to the close association between these two stages it is possible that they can be undertaken at the same time.
6. Product Recovery
Based on the outputs of the prior lab-scale Stages we can optimise the methods employed for separating and purifying the target product from the fermentation Stage. We can also potentially look at the recovery of other compounds from the broth.
It is possible for this Stage to run alongside Stage 5.
7. Valorisation of Remaining Biomass
Depending on the pretreatment, hydrolysis, and fermentation processess employed in the project, we can look at valorising those biomass components that are not hydrolysed to sugars and fermented to your target product.
For example, we can analyse the enzymatic hydrolysis residue for its suitability for combustion. In another scenario we can evaluate the potential for valorising the hemicellulose sugars that are hydrolysed in the dilute-acid-pretreatment stage.
8. Validation at Higher TRLs
Once we have concluded our optimisation of the SHF process conditions at the lab-scale we can then test those conditions at higher technology readiness levels (TRLs). The scales at which we can operate are dependent on the type of technology employed, but can reach up to 100 litres.
We have all of the necessary downstream equipment to efficiently handle the solid and liquid streams arising from these scaled-up activities.
If we find that there are differences between the yield and compositions of the different streams, compared with our lab-scale experiments, then we can explore the potential reasons for these and work on final tweaks to optimise the bioprocess for higher TRLs.
9. Technoeconomic Analysis (TEA)
The Celignis team, including Oscar our chief TEA expert, can undertake a detailed technoeconomic analysis of the developed process. We apply accurate and realistic costing models to determine the CAPEX and OPEX of simulated and pilot scale processes which are then used to determine key economic indicators such as IRR, NPV and payback periods.
Within these TEAs we can undertake sensitivity analyses to assess the effect of variable costs and revenues on the commercial viability of the process.
Our preferred approach is to include TEA studies at each stage of the development of the bioprocess, so that the process can be optimised in a commercially-relevant way, followed by a more detailed TEA after the process has been optimised and tested at higher TRL levels.
Click here to read more about the technoeconomic analysis (TEA) services offered by Celignis.
SHF Projects - Case Study
Bioethanol from Palm Residues
Celignis undertook a bioprocess development project for a client, based in the Middle East, that was targeting the production of bioethanol from the residues of local palm trees. This was a lab-scale vertically-integrated project covering pretreatment, and separate hydrolysis and fermentation (SHF).The client initially had a certain type of pretreatment technology in mind and requested that we undertake a series of experiments to assess it. However, based on our initial compositional analysis of the feedstocks, we had reservations that the chosen pretreatment would be suitable for such biomass. We discussed this with the client and it was agreed that three different types of pretreatments were tested, with each pretreatment type being undertaken a number of times in order to allow for an initial evaluation on the effects of varying the process parameters on the yield and compositions of the output streams.
The results from these initial pretreatment experiments confirmed Celignis's reservations regarding the originally-chosen pretreatment and resulted in the pretreatment technology that we recommended, based on the feedstock compositional data, being selected for further study.
There then followed a more extensive series of lab-scale experiments focused on optimising the pretreatment conditions so that the yields and commercial viability of the process as a whole could be improved. The next stage of the project then involved optimising the type and dosage of enzymes, as well as other factors such as the solid-loading, in order to maximise ethanol yields from the targeted biomass components.

Lalitha Gottumukkala
Founder of Celignis Bioprocess, CIO of Celignis
PhD
<p style="text-align: left;">Has a deep understanding of all biological and chemical aspects of bioproceses. Has developed Celignis into a renowned provider of bioprocess development services to a global network of clients.</p>

Oscar Bedzo
Bioprocess Project Manager & Technoeconomic Analysis Lead
PhD
<p style="text-align: left;">A dynamic, purpose-driven chemical engineer with expertise in bioprocess development, process design, simulation and techno-economic analysis over several years in the bioeconomy sector.</p>

Dan Hayes
Celignis CEO And Founder
PhD (Analytical Chemistry)
<p style="text-align: left;">Dreamer and achiever. Took Celignis from a concept in a research project to being the bioeconomy's premier provider of analytical and bioprocessing expertise.</p>