Enzymatic Hydrolysis Tests

Background on Enzymatic Hydrolysis
Enzymes are biocatalysts and play a key role in biorefinery processes for the biochemical/biological conversion of biomass to biofuels and biobased products. First generation and second generation biofuels use enzymes for conversion of structural polymers to monomers so that these can then be fermented to the desired fuel by yeast or bacteria.Starch Biorefinery Enzymes
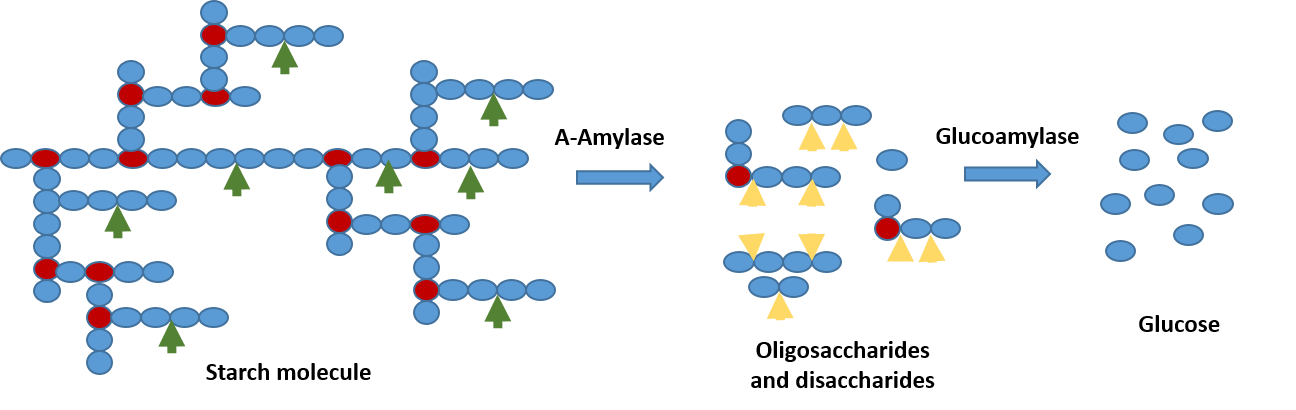
Amylases are key enzymes in starch biorefineries. There are two main kinds of amylases that are widely used in industrial applications:
|
Lignocellulose Biorefinery Enzymes
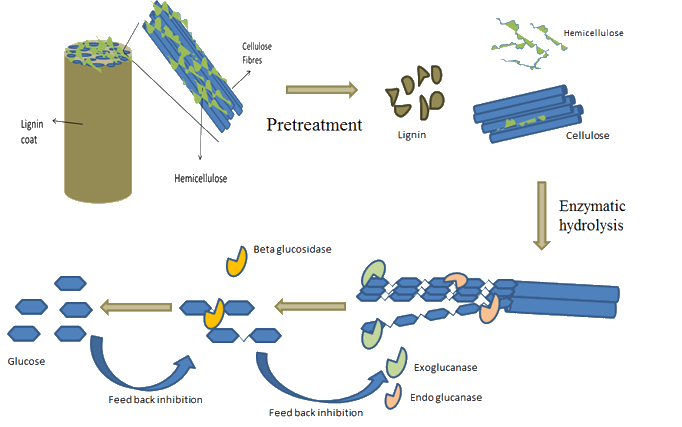
Plant biomass is a complex and recalcitrant matrix made of cellulose, hemicellulose and lignin. Lignocellulosic biomass typicaly needs a pre-treatment in order to make cellulose more accessible to enzymes.
The common class of enzymes that are used in lignocellulosic biorefineries are cellulases and xylanases. Lignin-degrading enzymes are also considered important in the biological and biochemical pre-treatment of biomass in order to avoid the formation of inhibitors.
Cellulase Enzymes
Cellulases are produced from both bacteria and fungi. Fungal cellulases are a complex of the following individual enzymes: Endoglucanase, exoglucanase and betaglucosidase. Fungal enzymes are the most commonly used cellulases in the advanced biofuels and biorefining industries because of the ability of fungi to produce these enzymes in high titres. The individual enzymes of cellulase act synergistically to breakdown cellulose to glucose:
|
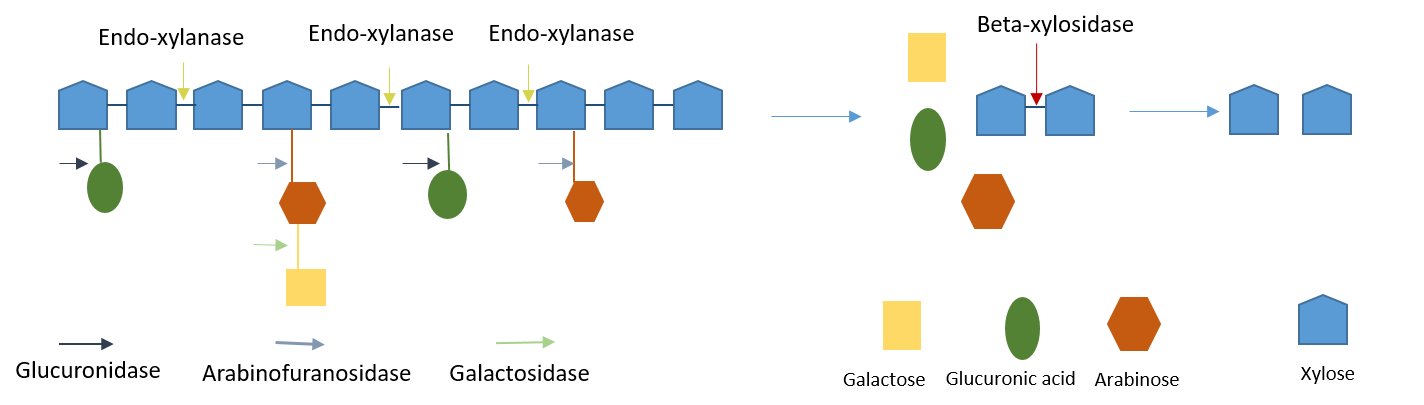
Xylanase Enzymes
Hemicellulose is a complex branched structure with different sugars and acidic compounds substituted at various positions on a backbone chain. As a result of this, multiple different enzymes are required for the complete hydrolysis of hemicellulose. Xylanases are mixtures of endoxylanases, beta-xylosidases, arabinofuranosidases, galactosidases, acetyl xylan esterases, and alpha-glucuronidases. The mix of xylanolytic enzymes act in synergy to convert xylan polymers to their monomeric constituents.
Endoxylanases reduce the viscosity of xylan by cleaving the xylan to soluble fragments and beta-xylosidases act on the soluble fragments and xylobiose to release xylose. Arabinofuranosidases and alpha-glucuronidases act on sugar and uronic-acid side groups of xylan.
Enzymatic Hydrolysis Tests at Celignis
We are pleased to offer a variety of analysis packages for evaluating both the suitability of biomass for enzymatic digestion and the activities of selected enzymes.Evaluation of Biomass and Pre-Treated Biomass for Enzymatic Digestibility
In analysis package P121 we add an enzyme solution to a sample and incubate it for 3 days, monitoring the sugars that are hydrolysed over time. Subsamples of the hydrolysate are collected after 0, 12, 24, 48, and 72 hours of digestion and are analysed for monosaccharides (glucose, xylose, mannose, arabinose, galactose, rhamnose) and cellobiose using our ion chromatography system. The resulting data allow us to plot a time-series of the concentrations of these sugars in solution and we also provide statistics for the conversion yield and conversion rate of cellulose and xylan.
These enzymatic hydrolysis tests can be carried out on native biomass and on pre-treated samples and we can either use a commercial enzyme mix or you can supply your own enzymes.
In order to carry out these tests we need to know the lignocellulosic composition of the sample so that we can use appropriate doses of enzymes and determine the conversion yields and rates. We recommend that analysis package P10 is used for determining this composition.
Evaluation of Pre-Treatment on Enzymatic Digestibility
Lignocellulose is a recalcitrant feedstock, with cellulose protected by lignin and hemicellulose, so making it less accessible to enzymatic degradation and microbial attack.
As a result, several pre-treatment strategies to increase the accessibility of cellulose and enhance the digestibility of lignocellulosic biomass by enzymes have been developed. An effective pre-treatment will result in increased accessibility of cellulose to enzymes and thus in high concentrations, yields and productivities of sugars in the enzymatic hydrolysate when compared to the un-treated biomass.
At Celignis we can undertake the enzymatic hydrolysis of a pre-treated sample using analysis package P121 or we can undertake a more detailed study to evaluate the efficacy of pre-treatment. This study involves analysis package P121 being undertaken on the native (non-pretreated) sample with analysis package P122 used for the enzymatic hydrolysis of the pre-treated sample(s). This package allows for the generation of a number of additional statistics outlining the relative advantages of the pre-treated sample(s) as substrates for enzymatic hydrolysis compared with the original sample. These statistics include:
|
|
Fermentation Inhibitors in Enzymatic Hydrolysate
It should be noted that an increase in digestibility does not always directly result in an increase in fermentability. Inhibitory sugar-degradation products including organic acids (e.g. acetic acid and formic acid), and furans (e.g. furfural and hydroxymethylfurfural) that may be formed during the pre-treatment process can be partially bound to the substrate and may be subsequently released into the enzymatic hydrolysate.
Based on their concentrations, these inhibitory compounds can negatively affect the fermentation process. For example, furans can inhibit cell replications at higher concentrations whilst weak acids can inhibit fermentation by accumulation of intracellular anions or by uncoupling of cell metabolism. Knowledge of the types of inhibitors in the enzyme hydrolysate and their concentrations will help in developing pre-treatment processes to avoid or reduce inhibitor formation and also to select suitable detoxification processes prior to fermentation.
We therefore recommend that the analysis of these potentially inhibitory compounds be undertaken when undertaking enzymatic hydrolysis tests, particularly those concerning the evaluation of the efficiency of biomass pre-treatment processes. Our analysis package P124 will determine the concentrations of these important inhibitors in the hydrolysate produced in our enzymatic hydrolysis tests.
P124 Fermentation Inhibitors in Enzymatic Hydrolysate |
|---|
| Formic Acid, Acetic Acid, Levulinic Acid, Furfural, Hydroxymethylfurfural |
Analysis of the Solid Residue after Enzymatic Hydrolysis
In order to get the most detailed information about the efficacy of enzymatic digestion, we recommend that the solid residue is analysed for its lignocellulosic composition. The resulting data will show what amounts of the structural polysaccharides have not been hydrolysed and will also help to inform decisions regarding how this residue is valorised.
In many biorefineries the solid residue after enzymatic hydrolysis is combusted or processed in other thermal conversion technologies (e.g. gasification or pyrolysis) in order to provide process heat and energy. If you would like to evaluate the hdyrolysis residue for this end-use then we recommend that analysis package P40 is also undertaken.
P40 Combustion Package |
|---|
| Volatile Matter, Fixed Carbon, Moisture, Ash, Carbon, Hydrogen, Nitrogen, Sulphur, Oxygen, Gross Calorific Value, Net Calorific Value, Chlorine |
Cellulase Saccharification Efficiency
At Celignis we also offer a range of analysis packages for evaluating enzymes and enzyme cocktails. In analysis package P125 we evaluate the efficiency of your enzymes for hydrolysing cellulose. The ability of cellulases to breakdown cellulose to its monomers is called saccharification efficiency and it can vary based on the activity of individual cellulase components. The efficiency is measured by determining the percentage of cellulose converted to glucose under standard/optimum conditions of the enzyme supplied.
Our cellulose saccharification test can be undertaken using pure cellulose or complex lignocellulosic substrates taken either from Celignis's repository or, alternatively, you can supply these substrates yourself.
Xylanase Saccharification Efficiency
The supplementation of xylanases with cellulases is an important strategy to improve the overall saccharification of lignocellulosic biomass. The ability of xylanase to hydrolyse the hemicellulose fraction of lignocellulose to xylose is called xylanase saccharification efficiency. It is measured by determining the percentage of xylan converted to xylose under standard/optimum conditions of the enzyme supplied. This test can be undertaken using pure xylan substrates or xylan-rich complex model substrates taken either from Celignis's repository or, alternatively, you can supply these substrates yourself.
Amylase Saccharification Efficiency
The efficiency of amylases to break down the starch polymer to its constituent monomeric sugars is called amylase saccharification efficiency. The level of efficiency and the extent of hydrolysis will depend on the activity of the enzymes alpha-amylase and glucoamylase in the amylase enzyme mix. In analysis package P127 the rate of hydrolysis of starch to glucose and percentage of starch converted to glucose are analysed and given in terms of conversion rate and conversion efficiency respectively.
Cellulase activity
We measure the activity of cellulase enzymes by filter paper assay (FPA) and express the activity in filter paper units (FPU/ml). The conversion of filter paper cellulose to glucose by cellulases is not a linear reaction. Hence, the assay requires enzyme dilutions to release fixed amount of glucose. Because the FPA is based on concentration of product released in a certain time in a nonlinear reaction, a conversion factor is used to approximate a specific activity-type unit.
P128 Cellulase Activity |
|---|
| Cellulase Activity |
Cellulolytic Enzymes Activity
As described earlier, cellulases are a complex of endocellulases, exocellulases and beta-glucosidases. The total activity of cellulase depends on individual enzymes activities and their ability to act in synergy. In analysis package P129 we can determine the activities of these indivifual enzymes, an important test that will help in understanding the limiting components in the enzyme mix. The different tests undertaken in this analysis package are outlined below:
|
P129 Cellulolytic Enzymes Activity |
|---|
| Cellulase Activity, Endoglucanase Activity, b-Glucosidase Activity, Xylanase Activity |
Amylolytic Enzymes Activity
With analysis package P130 we can determine the activity of amylolytic enzymes. This package involves the following tests:
|
P130 Amylolytic Enzymes Activity |
|---|
| a-Amylase Activity, Glucoamylase Activity |
Summary of Analysis Packages for Enzymatic Hydrolysis and Enzyme Activities
The analysis packages that we offer for enzymatic hydrolysis and enzyme activities, and the analytes that they determine, are listed below. Click on an entry for further details or go to the Celignis Database to place an order.
P124 Fermentation Inhibitors in Enzymatic Hydrolysate |
|---|
| Formic Acid, Acetic Acid, Levulinic Acid, Furfural, Hydroxymethylfurfural |
P128 Cellulase Activity |
|---|
| Cellulase Activity |
P129 Cellulolytic Enzymes Activity |
|---|
| Cellulase Activity, Endoglucanase Activity, b-Glucosidase Activity, Xylanase Activity |
P130 Amylolytic Enzymes Activity |
|---|
| a-Amylase Activity, Glucoamylase Activity |
Examples of Cellulosic Biomass Suitable for Enzymatic Hydrolysis Tests
Energy
Crops
Agricultural Residues and Wastes
Industrial Residues and Wastes
Municipal
Wastes
Biorefinery
Products






